Now researchers from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) in Berlin, in collaboration with colleagues from Milan, Paris and Mexico, have been able to highlight a new molecular mechanism: According to their discovery, the protein Rab35 and the mTOR signaling pathway it regulates play a central role in the formation of myelin sheaths in the peripheral nervous system. First in-vivo experiments show that new therapies can be derived from the findings. The work has now been published in the prestigious journal “Nature Communications”.
Many of our nerve cords (axons) are enveloped by a myelin sheath, which ensures that signals can be sent near instantaneously from the brain to muscles and organs. However, genetically programmed defects in myelination occur among the broad group of inherited neuropathies, disrupting this signaling process. This results in the onset of a variety of neurological deficits occurring in peripheral nerves and the degeneration of the nerve cords. This is the case with Charcot-Marie-Tooth disease (CMT), the most common inherited neuropathy. CMT type 4B is characterized by a very early onset of the disease; sufferers are often already confined to a wheelchair in their teens. In the worst case, neurodegeneration spreads to the respiratory tract, which can lead to death by respiratory failure. At present, there is no prospect of a cure.
Unexpected interaction partners
It is therefore all the more important to explore the largely unknown molecular mechanisms of the disease. This is exactly what scientists from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) in Berlin have done, in collaboration with teams of researchers led by Professor Alessandra Bolino (IRCCS Ospedale San Raffaele University, Milan), Professor Arnaud Echard (Sorbonne Université / Institut Pasteur, Paris) and Professor Genaro Patiño-López (Hospital Infantil, Mexico).
Researching the protein Rab35, the Berlin team led by Linda Sawade and Professor Volker Haucke discovered more or less by chance that this small GTPase, which is involved in the regulation of intracellular membrane transport, interacts with three proteins associated with CMT 4B: Owing to a gene mutation, MTMR2, MTMR5 and MTMR13 do not function properly in CMT 4B patients, or they are completely lacking.
These three critical proteins belong to the group of myotubularin-related (MTMR) phosphatidylinositol (PI) phosphatases that specifically hydrolyze the endosomal signaling lipids PI(3)P and PI(3,5)P2 at the 3’-phosphate group, i.e. they remove phosphates from lipids.
Rab35 regulates myelin sheath formation
“Our study revealed that the protein Rab35 regulates the longitudinal growth of the myelin sheath by binding and recruiting the two pseudophosphatases MTMR13 and MTMR5, and hence, also the active phosphatase MTMR2 bound to it in a complex,” reported Linda Sawade, lead author of the study.
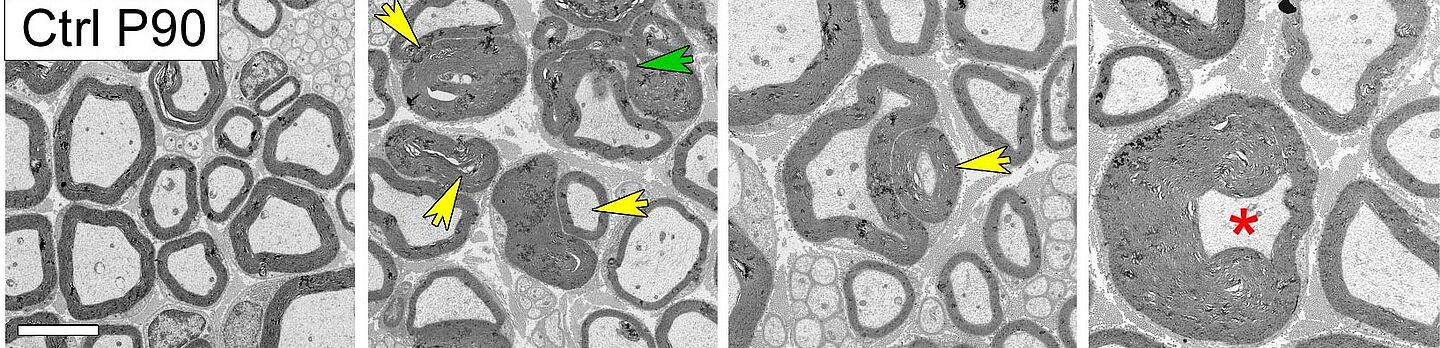
The new finding was that Rab35 binds this lipid phosphatase complex, and therefore plays a key role in regulating myelin sheath formation. The detection was confirmed in knock-out micethat specifically lack the Rab35 protein in Schwann cells – the cells in the peripheral nervous system that form myelin sheaths. Loss of the Rab35 protein led to the abnormalities and, eventually, the degenerative destruction (demyelination) of myelin sheaths in the sciatic nerve.
Inhibition of mTORC1 proves effective
Coincidentally, the researchers observed an abnormally elevated activity of the mTORC1 signaling pathway– one of the central signaling complexes for regulating myelin sheath formation in nerve tissue. Pharmacological inhibition of the hyperactive mTORC1 signaling complex using the drug Rapamycin partially rescued nerve damage in knock-out mice. Further experiments on cultured cells in which Rab35 expression had been suppressed confirmed the positive effects of mTORC1 inhibition on defective myelin sheaths.
The researchers were also able to draw an important conclusion from the absence of the Rab35 protein: mTORC1 is hyperactive because PI 3-phosphates are no longer regulated, causing the accumulation of PI(3)P and PI(3,5)P2 lipids. “We assume that this pathological process results from an impaired recruitment of MTMR complexes,” explained biochemist and cell biologist Linda Sawade. “Conversely, this would mean that Rab35 normally suppresses the activity of mTORC1 by recruiting MTMR phosphatases to lysosomes.”
Findings have an impact beyond basic research
In a nutshell, the results have a great impact for basic research: Rab35 is a previously unidentified regulator of myelin sheath formation in the peripheral nervous system and a repressor of mTORC1.
The results also offer a glimmer of hope to CMT4B patients: Therapeutic treatment using mTORC1-inhibiting drugs such as Rapamycin could improve disease progression. It would be the first treatment option for this serious condition.
Group leader Professor Dr. Volker Haucke: “Our work has led to the discovery of a new molecular mechanism in a particularly severe form of inherited neuropathy that is highly clinically relevant and that we now want to explore in greater depth with the Milan team led by Alessandra Bolino.”